Surfactants Making Personal Care and Homecare Easier

From cosmetics to hard surface cleaners, the consumers of the present world randomly search for naturally derived and safe products. Surfactants Suppliers India have been widely in demand for the distribution of surfactants to the industries for the manufacturing of a wide range of products such as personal care products, home care products, cosmetics, etc. The safe and simple formulations of natural surfactants have been widely accepted by many manufacturing industries due to their distinct properties which we will discuss in the coming sections of this blog post.
What are Surfactants?
Surfactants usually bring down the surface tension that arises in the liquid and gas, liquid and solid, or also two liquid and liquid interfaces. These chemical compounds have the features of wetting agents, emulsifiers, foaming agents, detergents, and dispersants. The term surfactant is derived from the phrase “surface-active agent”.
Types of surfactants
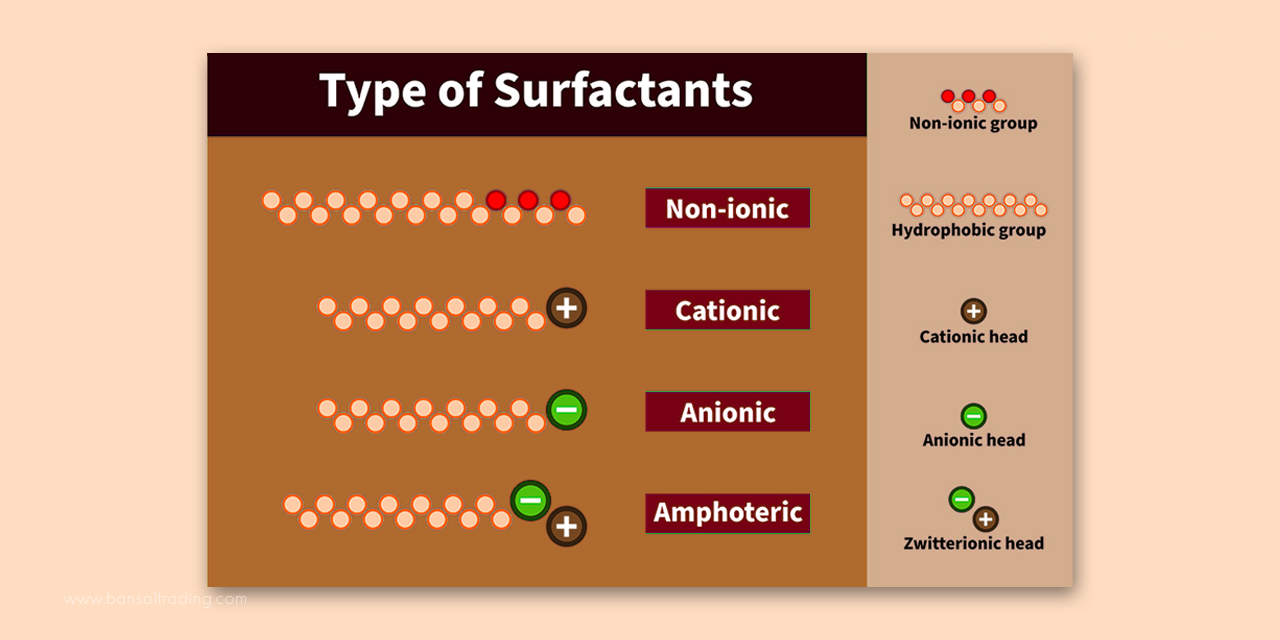
Surfactants can be divided based on the charge of the counter ion, or in terms of the formation of ions in the solution. They are known as amphoteric surfactants when exhibiting both positive and negative charges based on the pH levels of the solution. They are anionic surfactants when having a negatively charged ion. These are called cationic surfactants when they are charged positively. The non-ionic surfactants, as the name specifies, have zero charges. All these surfactants are widely used in shower products, premium shampoos, skin cleansers, personal cleansing products, Personal care products and cosmetics for various circumstances.
- Anionic Surfactants: They are the negatively charged ions and the core ingredients used in cleansing products. Some examples of anionic surfactants include sodium Lauryl Ether Sulphate, Sodium Lauroyl Sarcosinate, Sodium Cocoyl Isethionate, Sodium Lauroyl Glycinate, Ammonium Laureth Sulfate, etc. The chemical processing techniques of these surfactants make them more soluble and least irritating. The sulfosuccinates, acyl methyl laureates, alkyl benzene sulfonate, isethionates, monoglyceride sulfates, ether sulfonates, fatty glycerol, etc. are some other examples and the surfactant formula has been used in body washes, and shampoos. They eliminate dirt, and oil and produce soothing foam.
- Amphoteric Surfactants: The amphotericin surfactants exhibit both negative and positive charges based on the pH. They are also called zwitterionic materials. The ingredients of these surfactants include cocamidopropyl betaine, sodium lauraminopropionate, and cocoamphopropionate. These are best used in shampoos and other cleansing products. Compared to anionics, they have good emergency power and are less irritating. They make good foam, and make bubbles feel highly creamy and light.
- Nonionic surfactants: These are molecules that carry zero charges. Once placed in an aqueous solution, the molecules refuse to dissociate. Salt has no impact on chargeless surfactants. Some major types of fatty alkanolamides and fatty alcohols, cocamide DEA and Lauramide Diethanolamine, coco glucose, coco mono ethanol amide, and Decyl Glucoside are examples of non-ionic surfactants. Other ingredients like amine oxides, particularly stearamine oxide and lauramine oxides are found in cosmetics. They enhance foam while using anionics, prevent irritation, and give a soothing effect. They are also useful in solubilizing air fresheners, fragrances, and oils. Baby care products and shampoos are also nonionic, and the primary surfactants build emulsions.
- Cationic Surfactants: These surfactants are positively charged and not used for gentle cleansing properties since they don’t foam, clean, or rinse. However, they are the best surfactant examples for conditioning properties. They are an important addition to rinse-off hair conditioners.
Characteristics of Surfactants
- Detergency: There are two kinds of dirt on skin and hair— solid particles and oil deposits. The oil deposits are made in the hair follicles with natural sebum. The solid particles are collected from natural sources and remain on your skin/hair due to Van der Waals forces. Though rinsing the surface using water eliminates dirt, oily deposits stick onto the highly lipophilic parts of skin and hair. Surfactants present in detergents eliminate these oil deposits, where the lipophilic ends of molecules get aligned with the lipid layers on the hair and skin surface. Meanwhile, the lipid deposits get separated from the skin or hair, lift off, to finally wash away with the rinse.
- Wetting: Surfactants act as wetting agents that minimize the contact angle between solutions put on surfaces. This characteristic helps surfactants to spread easily across the surface and inject themselves along the oily deposit/ skin surface. This helps the product to get easily spread rather than balling up across the surface. This is an ideal formulation for creams and lotions.
- Foam: Foaming is an important feature in cleansing cosmetics. Foam is produced once the air gets dispersed in a continuous fluid medium. The air bubbles get wrapped with thin liquid layers, where the surfactants help to stabilize the formed bubbles. This eventually creates foam. However, note that the foam enhancers don’t serve much to the dirt removal process, but the consumers fall in love with the luxurious foam, for example in baby products, which makes it an essential factor for cleansing products to exhibit.
- Thickening: Surfactants form micelles—the water or surfactant solution usually has a majority of water, which they align into these structures. These small spherical structures activate the lipophilic tails inwards and the polar heads tend outwards the water. Since they are essential for thickening, they are also important for emulsion preparation. The thickness of the surfactant solution relies on the packing strength of micelles. The less proximity between micelles makes the solution much thinner. While the surface charge density reduces (adding salt), the particles get more closely packed, making the solution thicker.
- Emulsification: Another breakthrough of performance surfactants in cosmetics is the generation of semi-stable oil and water mixtures, and the creation of emulsions. Emulsions are lotions and creams that help the useful lipid materials get delivered over the skin and hair. They are either simply oil in water emulsions, water in oil emulsions, or sometimes complex multiple emulsions too.
Role of Surfactants in Home Care Products

- Cleaning products: The surfactants are highly used in home care solutions, industrial cleaners, and premium cleaning products. Almost 20-30% of the cleaning performance action comes with the ingredient of surfactants which makes an effective solution. They are used in liquid soaps, liquid hand soaps, mild cleansing products, etc.
- Shaving creams: Surfactants are also the best ingredients in lubricants like shaving creams, helping razors to easily eliminate stubble and avoid irritation.
- Car engine lubricants: They are used in the car engine lubricants to let the particles stick to engine parts, and let them move effectively, keeping the car in appropriate running condition.
- Detergents: Using surfactants in cleaning detergents helps them mix with water and get off the dirt from the surface to be cleaned. Surfactants make the cleaning process easier through effective mix-ups with water.
Also Read: What Are The Best Paint Raw Materials
Applications of Surfactants in Self-Care Products

Surfactants are the most significant among all cosmetic ingredients and personal care products. They have many useful applications, which proves their role in cosmetic and self-care products.
The major property of the molecules of surfactants is that they exhibit high-end compatibility with water and oil. While being put in the aqueous phase, they create structures naturally using lipid and hydrophilic alignment with water. The concentration of surfactant solution plays an integral role in the structure of cosmetics in micelles.
They are used as emulsifiers in the manufacturing of makeup products and skin care products. In the skincare routine, they can be used to make body washes, lotions, and creams. They are also ideal in gelling agents, thickeners, film-forming agents, etc. a
These are also great additions to personal care ingredients like hair care products, hair conditioner shampoo, keratin products, and hair relaxing treatment products.
- Creams & Lotions: In these personal care cosmetic products, you can use surfactants to soften and moisturize the skin. Lotions have greater water content than creams and have oil droplets suspended in an aqueous solution. Lotions are deeply absorbed in the skin and have less greasy properties. Creams have more oil content and that are also called water-in-oil emulsions with the water droplets suspended in an oil phase. Creams keep the skin hydrated. Emulsifiers should be less toxic, less irritating, and highly dispersible when choosing lotions and creams. PEG or Polyethylene glycols should be the base in these products with applications as emulsifiers or penetrating vehicles to help in skin lubrication.
- Body Wash: Being a key component in personal care cosmetic products, surfactant cleanses the skin with the effective elimination of lipophilic dirt and natural oils from your skin. It has a hydrophilic polar head and lipophilic nonpolar tail and assembles into spherical aggregates called micelles. The dirt and oil molecules get trapped in the micelle and the whole outer surface makes the micelle suspended in the water of the body wash. It gives more user experience while making fine foam to fill the wider surface area. Body washes generally vary in pH along the 6-7 scale. They are hence able to make good foam even while being acidic, with low irritation.
- Make-up Applications: Surfactants act as innovative ingredients like gelling agents, thickeners, and film-forming agents in beauty cosmetic & personal care products. They are the safe ingredients that enhance the volume, stability, and consistency of the product while also improving the water retention on the skin to play the role of moisturizers.
Also Read: Curious About What’s in Your Lipstick?
Conclusion
Since surface-active compounds minimize the tension between oil and water molecules, they are technically used to get rid of impurities, dirt, grease, etc. This is why cost-effective surfactant systems have been adopted widely by in-home care solutions and personal care applications making hygiene the key factor. Being one of the most trusted Surfactants Suppliers in India, Bansal Trading Company is destined to export and distribute a wide range of mild surfactants, chemicals, and raw materials to a wide variety of industries. Bansal Trading Company is a premier supplier of surfactants in India. Bansal Trading Company is the preferred choice for surfactants in India for personal care, home care, industrial applications, or specialty chemicals.
BTC_WEB_ADMIN






